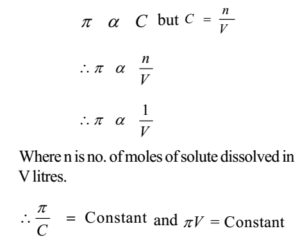The observed osmotic pressure for a 0.10M solution of Fe (NH4)2 (SO4)2 at 25^o C is 10.8 atm. The expected and experimental (observed) values of Van't Hoff factor (i) will be respectively: (

vant hoff equation for osmotic pressure of solution and expiration of degree of dissolution - Chemistry - - 14038313 | Meritnation.com

PPT - Osmotic pressure – van't Hoff equation : = g C R T Where: - osmotic pressure (atm or mm Hg) PowerPoint Presentation - ID:4239208

van't Hoff proved that osmotic pressure (pi) is a colligative property. For an ideal solution, osmotic pressure(pi) is helpful to determine that molecular mass of solute using M(B)=(W(B)RT)/(pi.V) Relation can exxpressed by

Osmosis/Osmotic Pressure/Vant Hoff Equation/Solutions/Explanation in Tamil/TN11th Std/CBSE 12 - YouTube